Exploring the distinction between positive and negative controls is vital in numerous domains, including scientific experimentation, clinical trials, and product evaluation. Grasping these concepts ensures the accuracy and reliability of experimental outcomes. This article delves into the definitions, applications, and implications of positive and negative controls, offering readers a deeper understanding of their significance in research and development. Through a detailed examination, this piece aims to equip you with the knowledge needed to differentiate between these two types of controls and appreciate their roles in experimental design.
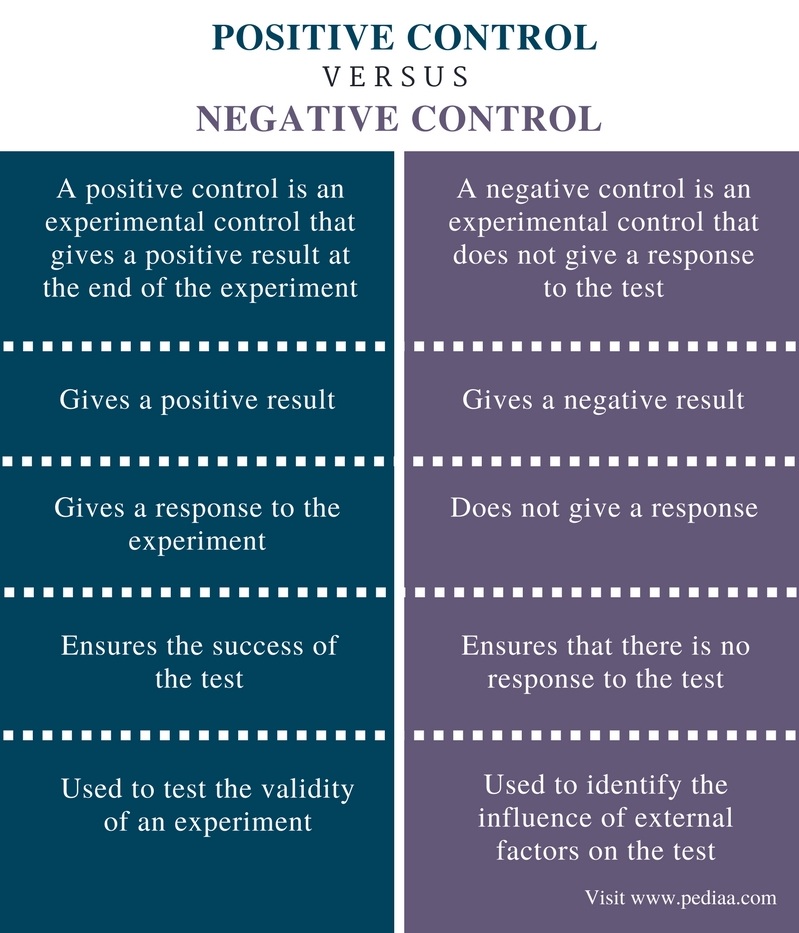
Positive control refers to a group within an experiment exposed to a treatment known to yield a positive outcome. It functions as a benchmark, confirming that the experimental setup is capable of delivering anticipated results. Conversely, negative control involves a group not exposed to the experimental treatment. This allows researchers to discern effects arising from external factors rather than the treatment itself, ensuring the validity of findings.
In the subsequent sections, we will explore the concepts of positive and negative controls in greater depth, discussing their importance, methodologies, and practical applications. By the conclusion of this article, readers will possess a clearer understanding of how these controls contribute to the credibility of scientific research.
Read also:Vincent Herbert A Multifaceted Music Maestro Who Redefined Excellence
Table of Contents
- Understanding Positive and Negative Controls
- The Importance of Positive and Negative Controls
- Implementing Controls in Experimental Design
- Applications of Positive and Negative Controls Across Fields
- Illustrative Examples in Real-World Research
- Challenges in Using Controls Effectively
- Best Practices for Control Implementation
- Summary and Final Thoughts
Understanding Positive and Negative Controls
Positive controls are groups or samples within an experiment subjected to a treatment known to produce a specific, desirable effect. For example, in a study evaluating drug efficacy, administering a well-established medication to a control group ensures that the experimental setup can reliably detect a positive response. This helps confirm the validity of the experimental design and the reliability of the results.
In contrast, negative controls involve groups or samples that do not receive the experimental treatment. Their primary role is to identify any false positives or external factors that might skew the results. For instance, in a clinical trial, a group receiving a placebo serves as a negative control, highlighting the effects of the absence of therapeutic intervention. This distinction is crucial for isolating the true impact of the treatment under investigation.
The Importance of Positive and Negative Controls
The role of positive and negative controls in research is indispensable. These controls play a critical part in:
- Ensuring Result Validation: Controls confirm that the experimental conditions are sufficient to generate reliable and accurate outcomes.
- Identifying Confounding Variables: They assist in isolating external factors that might influence the results, ensuring that observed effects are directly attributable to the treatment being tested.
- Improving Reproducibility: By incorporating controls, researchers enhance the ability to replicate experiments, bolstering the credibility and reliability of their findings.
Implementing Controls in Experimental Design
Successfully implementing positive and negative controls requires meticulous planning and attention to detail. The following steps outline the process:
- Clarifying Objectives: Clearly define the goals of the experiment to determine the most appropriate types of controls to include.
- Selecting Appropriate Groups: Choose representative samples for both positive and negative controls to ensure the validity of the results.
- Standardizing Procedures: Maintain consistent methodologies across all experimental groups to minimize variability and enhance reliability.
- Comprehensive Data Analysis: Analyze the results with controls in mind, comparing the outcomes of the experimental groups against the control groups to draw accurate conclusions.
Applications of Positive and Negative Controls Across Fields
Positive and negative controls find practical applications in a wide array of disciplines:
Clinical Trials
In clinical research, controls are indispensable for evaluating the effectiveness of new treatments. Positive controls help demonstrate that the treatment achieves the desired effect, while negative controls help identify placebo effects, ensuring that the observed outcomes are genuine.
Read also:Inspiring Life And Career Of Jamie Whitewelling
Environmental Testing
Environmental studies frequently employ controls to assess the impact of pollutants on ecosystems. Positive controls can confirm the presence of harmful substances, while negative controls help establish baseline conditions, ensuring that the results reflect the true impact of the pollutant.
Illustrative Examples in Real-World Research
Several noteworthy studies exemplify the practical use of positive and negative controls:
- Vaccine Trials: In vaccine development, positive controls may include existing vaccines, while negative controls involve placebo groups, ensuring that the new vaccine's efficacy is accurately measured.
- Drug Development: Pharmaceutical companies use positive controls to compare new drugs against established treatments, confirming their efficacy and safety before proceeding to market.
Challenges in Using Controls Effectively
While essential, implementing positive and negative controls presents several challenges:
- Selection Bias: Choosing inappropriate controls can lead to misleading or inaccurate results, compromising the validity of the study.
- Resource Constraints: Conducting comprehensive control studies often demands significant time, funding, and resources, which may not always be available.
- Complexity of Variables: In intricate biological systems, isolating the effects of the treatment from other variables can be challenging, requiring advanced methodologies and expertise.
Best Practices for Control Implementation
To enhance the reliability of experimental results, researchers should adhere to the following best practices:
- Ensure that the sample sizes for both control and experimental groups are adequate to support statistically significant conclusions.
- Maintain transparency in reporting control methodologies, allowing for peer review and replication of the study.
- Regularly review and update control protocols based on emerging research and advancements in the field.
Summary and Final Thoughts
In conclusion, understanding the distinctions between positive and negative controls is essential for conducting rigorous and reliable research. These controls not only validate experimental results but also enhance the credibility and trustworthiness of scientific findings. Researchers are encouraged to incorporate both types of controls into their studies to ensure accurate and dependable outcomes.
We invite you to share your thoughts and insights on this topic in the comments section below. Additionally, explore our other articles for further insights into scientific research practices and methodologies.
Thank you for reading, and we hope to engage with you again soon!